
EU MDR


Simplify EU MDR compliance with software for managing supplier data and documentation
Achieving EU MDR compliance under the EU Medical Device Regulation requires accurate supplier data, complete technical documentation, and consistent oversight across complex supply chains. Many organizations still rely on manual processes, making it difficult to keep data current, validate information, and stay audit-ready.
EU MDR compliance software helps centralize data, automate supplier engagement, and simplify documentation, enabling your team to reduce risk and improve visibility.
Core capabilities of our EU MDR solution:
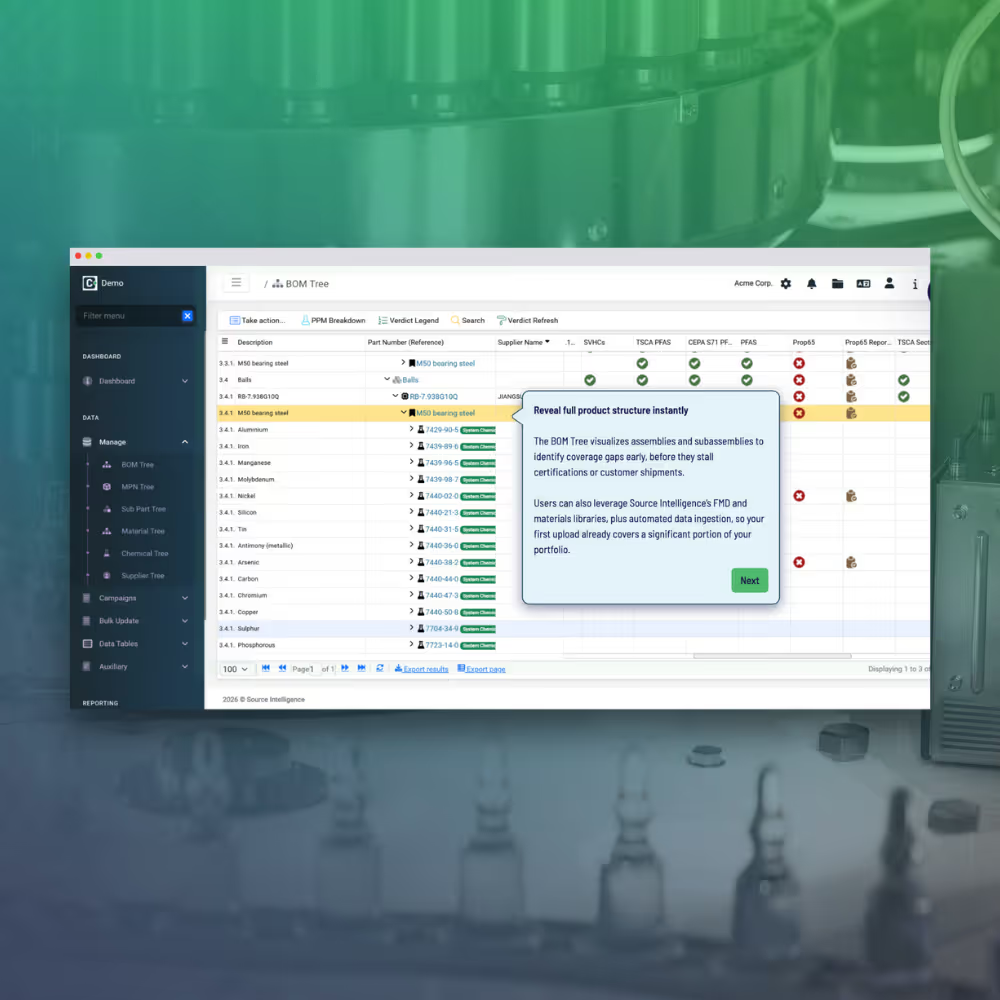
- Centralized compliance data across systems
- Pre-populated data from internal and external sources
- Real-time insights into compliance status and gaps
- Automated documentation for audit readiness
Simplify EU MDR compliance with software for managing supplier data and documentation
Achieving EU MDR compliance under the EU Medical Device Regulation requires accurate supplier data, complete technical documentation, and consistent oversight across complex supply chains. Many organizations still rely on manual processes, making it difficult to keep data current, validate information, and stay audit-ready.
EU MDR compliance software helps centralize data, automate supplier engagement, and simplify documentation, enabling your team to reduce risk and improve visibility.
Core capabilities of our EU MDR solution:
- Centralized compliance data across systems
- Pre-populated data from internal and external sources
- Real-time insights into compliance status and gaps
- Automated documentation for audit readiness


Understanding EU MDR requirements and compliance challenges
The EU Medical Device Regulation (EU MDR) introduces stricter requirements for data accuracy, technical documentation, and ongoing compliance oversight across the supply chain. Manufacturers must validate supplier data at the component level, maintain complete and up-to-date technical documentation, and ensure products meet evolving safety, labeling, and traceability expectations.
For many organizations, EU MDR compliance is difficult to scale. Supplier data is often incomplete or outdated, documentation is fragmented across systems, and manual processes slow response times. These challenges increase the risk of audit findings, delayed approvals, and loss of market access.
Understanding EU MDR requirements and compliance challenges
The EU Medical Device Regulation (EU MDR) introduces stricter requirements for data accuracy, technical documentation, and ongoing compliance oversight across the supply chain. Manufacturers must validate supplier data at the component level, maintain complete and up-to-date technical documentation, and ensure products meet evolving safety, labeling, and traceability expectations.
For many organizations, EU MDR compliance is difficult to scale. Supplier data is often incomplete or outdated, documentation is fragmented across systems, and manual processes slow response times. These challenges increase the risk of audit findings, delayed approvals, and loss of market access.
Risks of non-compliance with the EU MDR
When EU MDR compliance breaks down, the consequences extend beyond documentation gaps. Incomplete supplier data and missing technical records can quickly escalate into regulatory risk. This can disrupt operations and limit access to the European market, putting revenue and business continuity at stake.


EU MDR compliance software for efficient, scalable compliance management
Simplifying EU MDR compliance requires more than manual processes and fragmented systems. Teams need a centralized approach to collect, validate, and manage supplier data while maintaining complete, audit-ready documentation.
EU MDR compliance software from Source Intelligence helps organizations manage compliance at scale by connecting data across systems, reducing supplier outreach, and automating documentation workflows. By building data coverage first and engaging suppliers only where needed, teams can improve data quality, reduce response time, and maintain accurate records aligned with EU MDR requirements.
Our product compliance software helps teams scale EU MDR compliance efficiently without adding operational complexity.
How our EU MDR compliance software improves outcomes across your supply chain

Identify risks faster
AI-driven validation improves data accuracy and highlights gaps, helping teams address compliance issues earlier.

Improve operational efficiency
Automated workflows streamline supplier engagement and reduce manual effort across data collection and documentation.

Strengthen compliance confidence
Standardized data and centralized records support consistent, audit-ready documentation aligned with EU MDR requirements.

Increase visibility across the supply chain
Connected systems provide a clear view of compliance status, enabling better decision-making and faster response times.
Why choose Source Intelligence?

Educational resources

Understanding and Complying with EU MDR

Take Control: Medical Device Supply Chain Compliance

Managing Medical Device Regulatory Compliance

Software solutions to fit your needs




Simplify EU MDR compliance with software built for your supply chain

Events and news
See for yourself




