
EU MDR


Stay compliant with the EU Medical Device Regulation
Ensure your medical devices meet EU Medical Device Regulation (EU MDR) requirements with tools that streamline compliance. Our EU MDR solution makes it easier to work with suppliers, collect data, and create declarations. This helps businesses lower risk, stay compliant, and maintain market access in the European Union.
Core capabilities of our EU MDR solution:
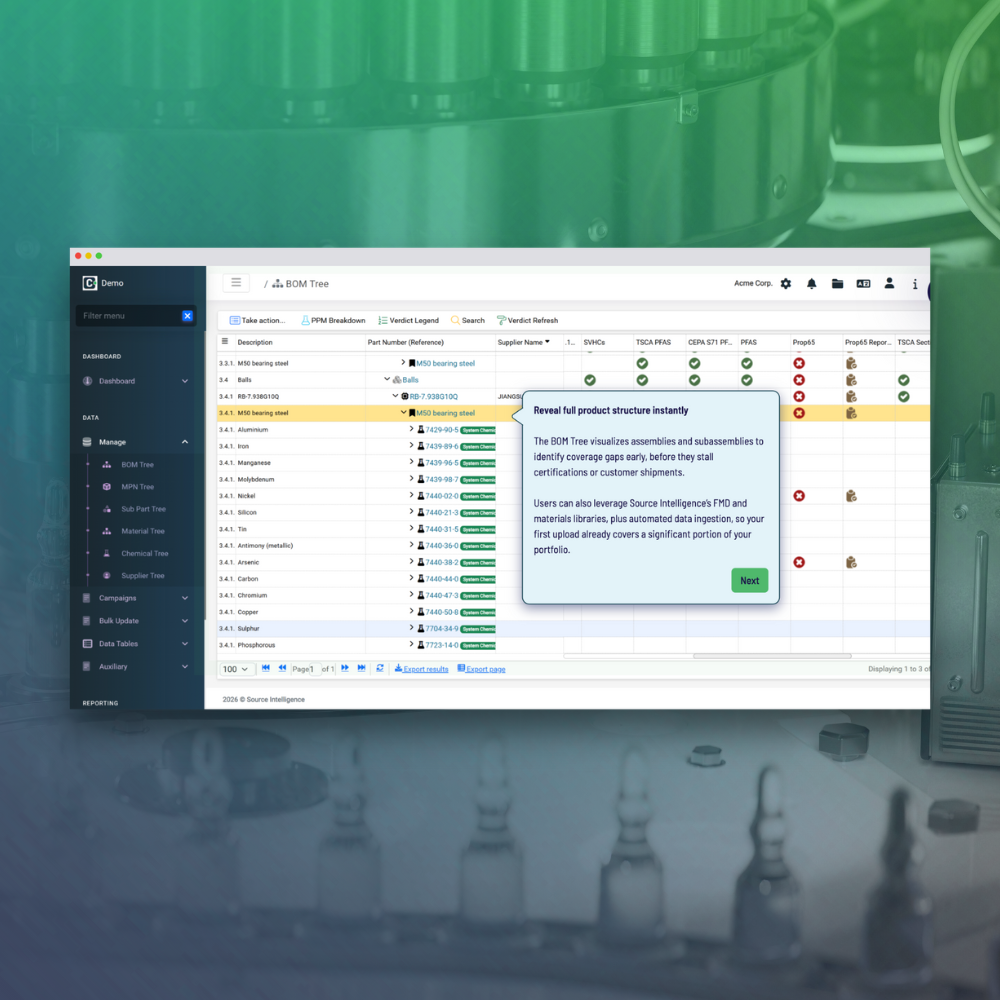
- Comprehensive data integration
- Streamlined supplier data collection
- Real-time compliance insights
- Effortless documentation generation
Stay compliant with the EU Medical Device Regulation
Ensure your medical devices meet EU Medical Device Regulation (EU MDR) requirements with tools that streamline compliance. Our EU MDR solution makes it easier to work with suppliers, collect data, and create declarations. This helps businesses lower risk, stay compliant, and maintain market access in the European Union.
Core capabilities of our EU MDR solution:
- Comprehensive data integration
- Streamlined supplier data collection
- Real-time compliance insights
- Effortless documentation generation


Navigating the EU MDR and compliance challenges
The EU MDR (2017/745) has strict rules for making, selling, and distributing medical devices in the European Union. The EU MDR replaces the Medical Devices Directive (MDD) and the Active Implantable Medical Devices Directive (AIMDD), improving safety, transparency, and quality standards. It requires manufacturers to monitor restricted substances, validate supplier data, and maintain detailed records, including EU MDR labeling requirements and documentation.
However, achieving compliance presents challenges for many businesses. Accurately tracking hazardous substances across every component can be complex. Managing supplier engagement, gathering compliance documentation, and meeting strict classification requirements stretch internal resources. Without the right tools, these demands increase the risk of penalties, product recalls, and reputational damage.
Navigating the EU MDR and compliance challenges
The EU MDR (2017/745) has strict rules for making, selling, and distributing medical devices in the European Union. The EU MDR replaces the Medical Devices Directive (MDD) and the Active Implantable Medical Devices Directive (AIMDD), improving safety, transparency, and quality standards. It requires manufacturers to monitor restricted substances, validate supplier data, and maintain detailed records, including EU MDR labeling requirements and documentation.
However, achieving compliance presents challenges for many businesses. Accurately tracking hazardous substances across every component can be complex. Managing supplier engagement, gathering compliance documentation, and meeting strict classification requirements stretch internal resources. Without the right tools, these demands increase the risk of penalties, product recalls, and reputational damage.
Risks of non-compliance with the EU MDR
Failure to comply with the EU regulation of medical devices can lead to serious consequences for manufacturers, including:


Simplify EU MDR compliance with Source Intelligence
Our EU MDR solution addresses the complexities of EU MDR compliance with tools that automate supplier engagement, streamline data collection, and ensure reporting accuracy. Key features include centralizing and validating supplier information to generate technical documentation and meet EU MDR labeling requirements, including the CE marking. Whether you prefer a self-service solution or need managed services, our platform helps businesses meet compliance goals easily.
How our EU MDR solution strengthens your compliance processes

Identify risks faster
Artificial intelligence (AI) capabilities improve data quality, delivering insights into critical risk areas and improving supply chain visibility.

Boost efficiency
Automated workflows streamline supplier outreach, and automated documentation generation minimizes manual work.

Mitigate compliance risks
Standardized data collection templates, developed by regulatory experts, ensure compliance data is reliable and actionable.

Increase risk foresight
Our regulatory experts guide our solution development, enabling you to prepare for regulatory changes.
Why choose Source Intelligence?

Educational resources

EU MDR Compliance: A Complete Guide

Product Compliance for Medical Device Manufacturing

6 Best Practices for Product Compliance Management

Software solutions to fit your needs




Streamline compliance with our EU MDR solution

Events and news
See for yourself




