
Global REACH


Mitigate risks and ensure REACH compliance with confidence
Proactively manage your REACH compliance obligations with tools designed to identify and mitigate risks associated with hazardous substances. Our global REACH solution leverages artificial intelligence and automation to help you achieve compliance. Ensure your products meet REACH reporting requirements, reduce risks to human health and the environment, and maintain market access.
Core capabilities of our Global REACH solution:
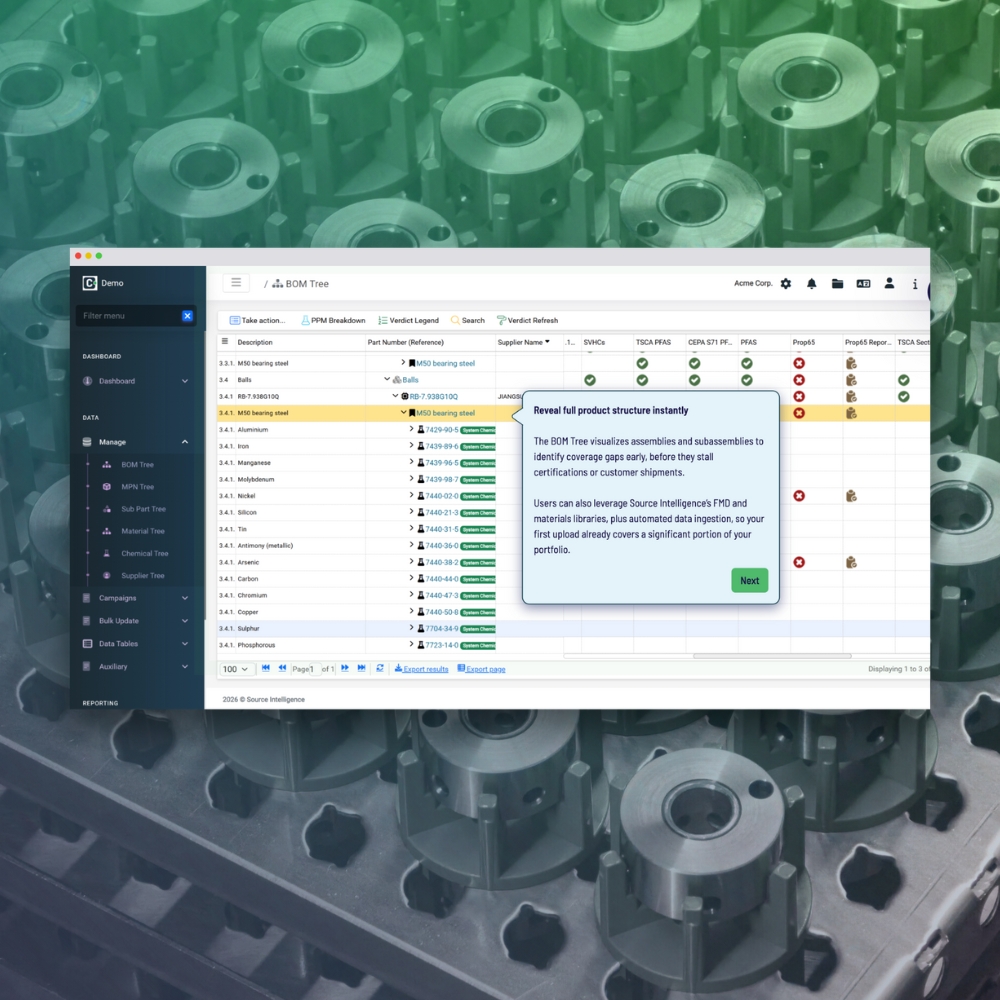
- Comprehensive data integration
- Streamlined supplier data collection
- Real-time compliance insights
- Effortless declaration generation
Mitigate risks and ensure REACH compliance with confidence
Proactively manage your REACH compliance obligations with tools designed to identify and mitigate risks associated with hazardous substances. Our global REACH solution leverages artificial intelligence and automation to help you achieve compliance. Ensure your products meet REACH reporting requirements, reduce risks to human health and the environment, and maintain market access.
Core capabilities of our Global REACH solution:
- Comprehensive data integration
- Streamlined supplier data collection
- Real-time compliance insights
- Effortless declaration generation


Navigating REACH regulatory compliance
REACH stands for The Registration, Evaluation, Authorization, and Restriction of Chemicals. The global regulatory framework restricts the use of harmful substances in products to protect human health and the environment. Implemented in regions like the European Union and the United Kingdom, REACH presents varying compliance requirements that manufacturers and importers must navigate.
Ensuring compliance across markets involves validating supplier information and gathering essential documentation, including FMDs and REACH compliance certificates. Businesses must also stay updated on evolving regulatory lists, such as the EU REACH Regulation’s Substances of Very High Concern (SVHC) list. These processes demand significant resources, often leaving businesses vulnerable to non-compliance.
Navigating REACH regulatory compliance
REACH stands for The Registration, Evaluation, Authorization, and Restriction of Chemicals. The global regulatory framework restricts the use of harmful substances in products to protect human health and the environment. Implemented in regions like the European Union and the United Kingdom, REACH presents varying compliance requirements that manufacturers and importers must navigate.
Ensuring compliance across markets involves validating supplier information and gathering essential documentation, including FMDs and REACH compliance certificates. Businesses must also stay updated on evolving regulatory lists, such as the EU REACH Regulation’s Substances of Very High Concern (SVHC) list. These processes demand significant resources, often leaving businesses vulnerable to non-compliance.
Risks of REACH non-compliance
Non-compliance with REACH regulations can lead to significant penalties, legal consequences, and operational challenges, including:


Streamline Global REACH compliance with Source Intelligence
Source Intelligence’s Global REACH solution simplifies compliance by automating supplier engagement, data collection, and documentation generation. Our platform facilitates the efficient gathering and validation of necessary data for REACH reporting, including declarations, FMDs, and lab test results. We tailor our solution to your business needs, offering software and managed services to ensure seamless compliance.
How our Global REACH solution drives compliance success

Identify risks faster:
Artificial intelligence (AI) capabilities improve data quality, delivering insights into critical risk areas and improving supply chain visibility.

Boost efficiency:
Automated workflows streamline supplier outreach, and automated documentation generation minimizes manual work.

Mitigate compliance risks:
Standardized data collection templates, developed by regulatory experts, ensure compliance data is reliable and actionable.

Respond quickly to requests:
Generate REACH compliance statements and declarations on demand to meet customer and regulatory requests efficiently.
Why choose Source Intelligence?

Educational resources

Understanding REACH on a Global Scale

Five Step Guide to EU REACH Compliance

EU REACH vs. RoHS Compliance: What is the Difference?

Software solutions to fit your needs




See how effortless REACH compliance can be with our compliance solutions.

Events and news
See for yourself




